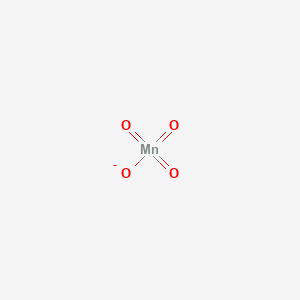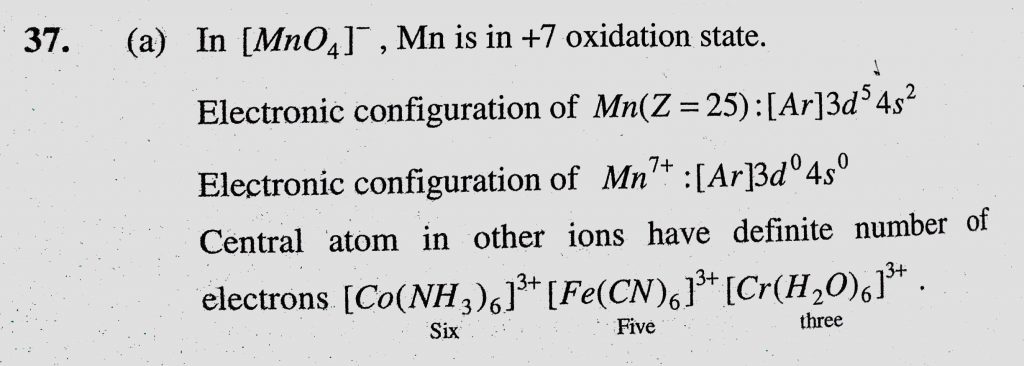
The complex ion which has no 'd' electrons in the central metal atom is (a) (MnO4)- (b) (Co(NH3)6) 3+ - Sahay Sir
Solved] Give the name of Ca(MnO4)2 and calculate its molar mass. 2. What is the volume, in nm3 , of a wooden block with a volume of 43.7 cm3? 3. Wha... | Course Hero

If S + O2→ SO2, H = - 298.2 kJ mole^-1 SO2 + 1/2 O2→ SO3, H = - 98.7 kJ mole^-1 SO3 + H2O → H2SO4, H = - 130.2 kJ

How hydrogen‐bonded MnO4‐ can influence oxidation of olefins in both gas phase and solution? - Javan - 2012 - Journal of Physical Organic Chemistry - Wiley Online Library

SOLVED:What is the xidation number change (final- initial) for the manganese atom in the following unbalanced reduction half reaction? MnO4 - (aq) + Ht(aq) + Mn2+( (aq) + HzO() O A. -5

How hydrogen‐bonded MnO4‐ can influence oxidation of olefins in both gas phase and solution? - Javan - 2012 - Journal of Physical Organic Chemistry - Wiley Online Library











![MnO4]2- - Manganate MnO4]2- - Manganate](https://www.chemtube3d.com/images/gallery/inorganicsjpgs/MnO42-.jpg)